Observing Mitosis with Fluorescence Microscopy
Mitosis, a phenomenon observed in all higher eukaryotes, is the mechanism that allows the nuclei of cells to split and provide each daughter cell with a complete set of chromosomes during cellular division. This, coupled with cytokinesis (division of the cytoplasm), occurs in all multicellular plants and animals to permit growth of the organism. Digital imaging with fluorescence microscopy is becoming a powerful tool to assist scientists in understanding the complex process of mitosis on both a structural and functional level.
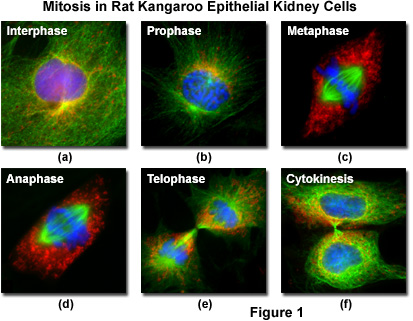
Fluorescent probes can be attached to specific subcellular components, such as the chromosomes and microtubules, for visualization of mitosis using standard epi-fluorescence microscopy techniques. The technology relies on the fact that fluorescent molecules absorb light at one wavelength and emit secondary (fluorescence) light at a longer wavelength. By employing a carefully selected combination of filters, the short wavelength light can be removed from the imaging pathway to produce strikingly beautiful images of the bright structures superimposed on a black background, as illustrated in Figure 1 for several important stages in mitosis.
Living epithelial kidney cells, derived from the rat kangaroo (Potorous tridactylus) and grown in culture, are often used to visualize mitosis in the microscope because they contain only a few large chromosomes and the cells remain relatively flat throughout all of the division stages. Termed PtK2, the marsupial kidney cells afford clear visualization of the chromosomes, mitotic spindle, nucleoli, and other components during mitosis. At 37 degrees Celsius, PtK2 cells undergo mitosis in approximately 2 to 3.5 hours and a typical healthy, growing culture can easily contain cells at every stage. Figure 1 presents images from five of the mitosis stages, as well as the interphase period. The cellular DNA was stained with DAPI (blue), while the mitochondria were stained with MitoTracker Red CMXRos (red) and the microtubules stained green with Alexa Fluor 488 attached to secondary antibodies.
Although the process of mitosis is often segregated, for descriptive purposes, into four or more discrete phases based on the appearance and behavior of chromosomes, the entire cell cycle is a dynamic event that occurs continuously without pause. For this reason, it should be borne in mind that the phases of mitosis are arbitrary designations and serve only as a tool for studying and describing the entire process of cell division.
Phases of Mitosis
Interphase - Between mitotic divisions, a normal resting or actively growing cell exists in a state known as interphase, in which the chromatin forms a highly diffuse, fibrous network that is being continuously transcribed by enzymes within the nucleus (Figure 1(a)). Before the cell enters the mitosis sequence, it first undergoes a DNA synthesis (or S) phase where each chromosome is duplicated to produce an identical pair of sister chromatids joined together by a specific DNA sequence known as a centromere. Centromeres are crucial to segregation of the daughter chromatids during mitosis.
Prophase - The first stage of mitosis is known as prophase, where the nuclear chromatin starts to become organized and condenses into thick strands that eventually become chromosomes observable in the optical microscope (Figure 1(b)). The nucleoli, primarily responsible for the production of ribosomal RNA, begin to disappear as the chromosomes condense. During prophase, major changes also occur in the cytoplasm, including disassembly of the cytoskeleton components based on tubulin (cytoplasmic microtubules). The tubulin monomers are dynamically redirected by the dividing cell to form the main component of the mitotic apparatus, the mitotic spindle, which is bounded by the centrosomes and begins to appear along the periphery of the nuclear membrane.
Prometaphase - Late prophase, or prometaphase, begins with the disruption of the nuclear envelope, which is broken down into small membrane vesicles that closely resemble the endoplasmic reticulum and tend to remain visible around the mitotic spindle. During this period the chromosomes continue to condense and gradually shorten and thicken until they have completely formed the units that will undergo mitosis. The nucleolus, which may still be present in some cells, also completely disappears in prometaphase. In the absence of the nuclear membrane, the mitotic spindle microtubules are now free to enter the nuclear region, and formation of specialized protein complexes called kinetochores begins on each centromere. These complexes become attached to some of the spindle microtubules, which are then termed kinetochore microtubules. Other microtubules in the spindle (not attached to centromeres) are termed polar microtubules and these help form and maintain the spindle structure along with astral microtubules, which remain outside the spindle.
Metaphase - Perhaps the most recognizable phase of mitosis is termed metaphase, a stage where the chromosomes, attached to the kinetochore microtubules, begin to align in a single plane (known as the metaphase plate) midway between the spindle poles (see Figure 1(c)). The kinetochore microtubules exert tension on the chromosomes, which move back and forth in rapid erratic motion as a result, and the entire spindle-chromosome complex is now ready for the next event, separation of the daughter chromatids. Metaphase, one of the most critical stages in mitosis, occupies a substantial portion of the division cycle. The primary reason for this extended interval is that dividing cells pause until all of their chromosomes are completely aligned at the metaphase plate. This sets the stage for chromosome separation in the next stage of mitosis, termed anaphase.
Anaphase - Almost immediately after the metaphase chromosomes are aligned at the metaphase plate, the two halves of each chromosome are pulled apart by the spindle apparatus and migrate to the opposite spindle poles (Figure 1(d)). The kinetochore microtubules shorten as the chromosomes are pulled toward the poles, while the polar microtubules elongate to assist in the separation. Anaphase typically is a rapid process that lasts only a few minutes. When the chromosomes have completely migrated to the spindle poles, the kinetochore microtubules begin to disappear, although the polar microtubules continue to elongate. This is the junction between late anaphase and early telophase, the last stage in chromosome division. In photomicrographs of the process, polar microtubules are in a clearly formed network and the synthesis of a new cell membrane is initiated in the cytoplasm between the two spindle poles.
Telophase - In telophase, the daughter chromosomes arrive at the spindle poles and are eventually redistributed into chromatin. The process of cytokinesis, where the cytoplasm is divided by cleavage, also starts sometime in late anaphase and continues through telophase (see Figure 1(e)). After complete separation of the chromosomes and their extrusion to the spindle poles, the nuclear membrane begins to reform around each group of chromosomes at the opposite ends of the cell. The nucleoli also reappear in what will eventually become the two new cell nuclei. When telophase is complete and the new cell membrane (or cell wall in the case of higher plants) is being formed, the nuclei have almost matured to the pre-mitotic state. The final steps in telophase involve the initiation of plasma membrane cleavage between each of the new daughter cells to ultimately yield two separate cells during cytokinesis, the next phase of cell division.
Cytokinesis - The final stage in the process of cell division is known as cytokinesis, which usually begins during late anaphase or early telophase (before mitosis ends) as the nuclear envelope and nucleoli are reforming and the chromosomes are de-condensing (see Figure 1(f)). During cytokinesis, the cytoplasm divides by a process termed cleavage, driven by the tightening of a contractile ring composed of actin and myosin protein subunits. As the ring of cytoskeletal proteins contracts, a cleavage furrow is formed perpendicular to the mitotic spindle and gradually splits the cytoplasm and its contents into two daughter cells.
Contributing Authors
John D. Griffin, Nathan S. Claxton, and Michael W. Davidson - National High Magnetic Field Laboratory, 1800 East Paul Dirac Dr., The Florida State University, Tallahassee, Florida, 32310.
