Fluorescence Digital Image Gallery
Normal Rat Kidney Epithelial Cells (NRK Line)
The NRK cell line was derived from the kidney of a rat of the species Rattus norvegicus. The cells grow adherently and exhibit epithelial morphology. Epithelial cells are cells that exist in the body in sheets covering the organs and other internal and external surfaces that may come into contact with foreign materials.
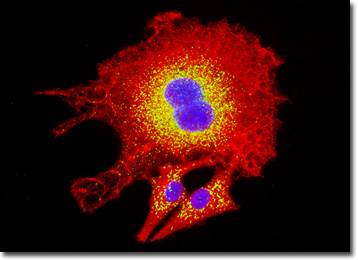
Typically epithelial cells contain a relatively large amount of cytoplasm and a significant quantity of granules. The function of the cells is various, some acting in an absorptive or protective role, while others primarily act as secretory cells. The epithelial cells of the kidneys chiefly fall into this latter category, playing an important role in the storage and subsequent secretion of various excretory materials.
In addition to various functions, epithelial cells may exhibit a number of different shapes depending upon where they are located in the body. The principal types of the cells include columnar, cuboidal, squamous, and ciliated varieties. Columnar epithelial cells, which are elongate and somewhat cylindrical, are chiefly found lining the intestinal tract as well as along the ducts of most glands. In contrast, the cuboidal-type epithelial cells, which as their name implies are cubical in shape, are found on the surface of the brain, in the middle ear, and lining glands and ducts, including those of the kidneys. Squamous epithelial cells are thin and plate-like. Typical areas of the body where they are located include the alveoli of the lungs and the glomeruli and capsules of the kidneys. Epithelial cells that feature thin hair-like projections are said to be ciliated. Ciliated epithelial cells occur in the bronchi of the lungs, certain areas of the nasal cavities, and in the reproductive systems of both males and females.
The culture of normal rat kidney cells presented in the digital image above was immunofluorescently labeled with primary anti-clathrin (heavy chain) mouse monoclonal antibodies followed by goat anti-mouse Fab fragments conjugated to Texas Red in order to target the cytoskeletal network. Additionally, peroxisomes present in the culture were immunofluorescently labeled with Alexa Fluor 488 conjugated to goat secondary antibodies directed against rabbit anti-PMP 70 (peroxisomal membrane protein 70). Nuclei were counterstained with Hoechst 33342. Images were recorded in grayscale with a QImaging Retiga Fast-EXi camera system coupled to an Olympus BX-51 microscope equipped with bandpass emission fluorescence filter optical blocks provided by Omega Optical. During the processing stage, individual image channels were pseudocolored with RGB values corresponding to each of the fluorophore emission spectral profiles.
Additional Fluorescence Images of Normal Rat Kidney Epithelial (NRK) Cells
Microtubular Network and Golgi Complex in NRK Cells - The microtubules present in a culture of normal rat kidney cells were immunofluorescently labeled with primary anti-tubulin (pan) mouse monoclonal antibodies followed by goat anti-mouse Fab fragments conjugated to Alexa Fluor 488. Golgi bodies were simultaneously targeted with rabbit anti-giantin primary antibodies, followed by goat anti-rabbit secondaries conjugated to Texas Red. The ultraviolet-absorbing probe Hoechst 33342 was utilized to counterstain nuclei.
Normal Rat Kidney Cells with MitoTracker Red CMXRos, Alexa Fluor 488, and DAPI - The rat kidney epithelial cells presented in this section were resident in a culture stained with MitoTracker Red CMXRos, Alexa Fluor 488 conjugated to phalloidin, and DAPI, targeting mitochondria, the cytoskeletal filamentous actin network, and nuclear DNA, respectively.
Filamentous Actin in Rat Kidney Epithelial Cell Cultures - The F-actin present in a culture of NRK epithelial cells was targeted with Alexa Fluor 350 conjugated to phalloidin, a bicyclic peptide isolated from the death cap mushroom (Amanita phalloides). In addition, the culture was labeled for mitochondria with MitoTracker Red CMXRos, and for nuclei with SYTOX Green.
BACK TO THE CULTURED CELLS FLUORESCENCE GALLERY
BACK TO THE FLUORESCENCE GALLERY
