Nuclear Pores
The nuclear envelope is perforated with tiny holes known as nuclear pores, which were first discovered in the mid-twentieth century. These pores regulate the passage of molecules between the nucleus and cytoplasm, permitting some to pass through the membrane, but not others. Building blocks for manufacturing DNA and RNA are some of the materials that are allowed into the nucleus, as are molecules that provide the energy for constructing genetic material. Ribosomal subunits, which are built in nucleosomes, are a prime example of materials that must be allowed to leave the nucleus and enter the cytoplasm.
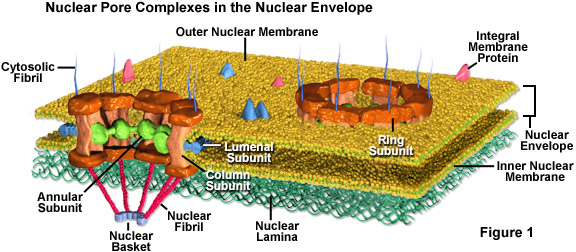
Nuclear pores are fully permeable to small molecules up to the size of the smallest proteins, but form a barrier keeping most large molecules out of the nucleus. Yet, some larger proteins, such as histones, are granted admission into the nucleus despite the fact that that the pores should be too small to let them through. It is generally thought that the elaborate protein structure called the nuclear pore complex (see Figure 1) that surrounds each pore plays a key role in allowing the active transport of a select set of large molecules into and out of the nucleus.
The nuclear pore complex is comprised of several subunits. Surrounding the inside of the pore is a nonmembranous material organized into an annulus that extends spoke-like structures toward the center of the small channel. The actual pore wall is predominantly comprised of columnar subunits, and lumenal subunits, with the help of transmembrane proteins, anchor the entire pore complex into the nuclear envelope. Also, tiny fibrils usually extend form both sides of the complex and congregate into basket-like configurations on the nuclear side of the complex. The proteinaceous composition of these fibrils is different on opposing sides of the structure.
In addition to their role in nuclear transport, nuclear pores are important as sites where the outer membrane and inner membrane of the nuclear envelope are fused together. Due to this fusion, the membranes can be considered continuous with one another although they have different biochemical characteristics and can function in distinctive ways. Since the outer nuclear membrane is also continuous with the membrane of the endoplasmic reticulum (ER), both it and the inner nuclear membrane can exchange membranous materials with the ER. This capability enables the nuclear envelope to grow bigger or smaller when necessary to accommodate the dynamic contents of the nucleus.
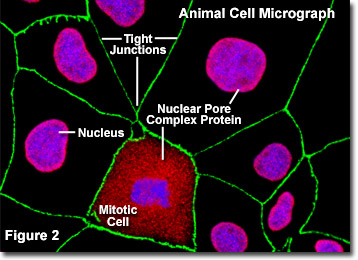
Illustrated in Figure 2 is a fluorescence digital image of an adherent culture of Madin-Darby canine kidney cells (MDCK line) stained with fluorescent probes targeting the nucleus (blue), nuclear pore complex proteins (red), and the tight junctions formed between epithelial cells (green) to demonstrate the proximity of these structures. The nuclear pores of these cells were targeted with a wide-spectrum polyclonal antibody to a large family of nuclear pore complex proteins, which serves as a useful tool for studying the morphology and composition of the nucleus and nuclear envelope. The antibody mixture is also useful in studying changes in the nuclear structure during mitosis and meiosis (note the mitotic cell in the lower central portion of Figure 2).
Pore density varies greatly and tends to be greatest among cells that are highly activate and differentiated, such as liver cells. A typical mammalian cell features approximately 3,000 to 4,000 pores along its nuclear envelope. The oocytes of certain amphibians, however, have such large nuclei and such a density of pores that the nuclear envelope of one of the cells may contain more than ten million pores. Consequently, these oocytes have been heavily utilized in studies of nuclear pore complexes and nuclear transport.
