Total Internal Reflection Fluorescence Microscopy
Olympus Application Note
Total internal reflection microscopy (TIRFM) is an optical technique used to observe single molecule fluorescence. Some biophysicists have used the technique for many years, while others are just beginning to explore the boundaries of this versatile mechanism for studying phenomena occurring at interfaces.
Today, the technique is gaining popularity with cell biologists and neuroscientists to employ it to observe cell membrane fluorescence, in part because new membrane-specific dyes have been developed. In the past, TIRFM was difficult to perform due to the complexity of the microscope setup and the problem of achieving acceptable image brightness. A recently developed high numerical aperture microscope objective lens that improves TIRFM and makes it widely accessible is discussed in this application note.
Background
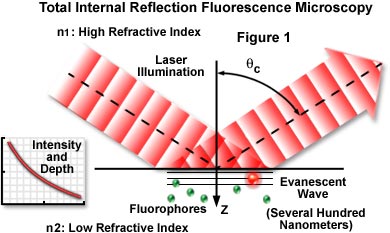
Total internal reflection is an optical phenomenon that can be employed to observe events occuring at boundaries. When light strikes the interface between two optical media of different refractive indices, the light incident at an angle greater than the critical angle undergoes total reflection.

Beyond the angle of total reflection, the electromagnetic field of the incoming/reflected light still extends into the z direction. The strength of this field, often termed the evanescent wave, decreases exponentially, and its effects extend only a few hundred nanometers into the second medium (having the lower refractive index). That portion of the specimen within the evanescent field can be excited to emit fluorescence and consequently can be seen or recorded. This is the essence of TIRFM (see Figure 1).
The condition for total reflection is given by the following equation:
where n(1) is the refractive index of the objective, n(2) is the refractive index of the specimen, and q(c) is the critical angle. This condition is identical to that creating the evanescent wave.
The key advantage of TIRFM is the shallow penetration depth of the evanescent wave. Only fluorophore molecules very near the surface of a specimen are excited to emit, creating an extremely thin optical section. Outside the evanescent field, fluorescence is minimal. This (see Figure 2) effect leads to images of very high contrast having a good signal-to-noise ratio.
Instrumental Approaches to TIRFM
Scientists have developed several TIRFM setups, of which the following types are common: (1) with illumination side prism, and (2) through the lens illumination (see Figure 3).
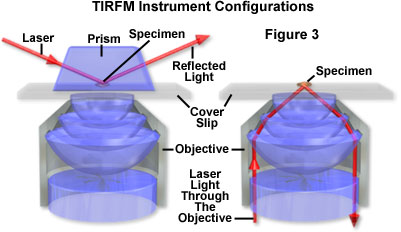
1. Setup with prism. This setup is easily accomplished, since it requires only the microscope, prism, and laser, all components that are readily available. The drawback for this setup is the requirement that the specimen be positioned between the prism and the microscope objective.
2. Setup through the objective lens illumination. This setup requires that the laser be introduced through the microscope, and greatly benefits from an objective lens with a numerical aperture (NA) > 1.4. The specimen must be located on the bottom surface of the cover glass. The preparation, facing away from the objective, is accessible and permits the use of other instrumentation such as micromanipulators, differential interference contrast (DIC) illuminating optics, and even a scanning probe microscope.
TIRFM Objective
The numerical aperture of the Olympus APO 100x/NA 1.65 apochromatic objective lens (Olympus America Inc., Melville, NY) greatly exceeds that of the previously highest numerical aperture objective lens, i.e., the Plan APO 100x/NA 1.4 (Olympus America Inc.). Living cells typically have a refractive index between 1.33 and 1.38. To achieve total internal reflection, such a specimen must be illuminated with an numerical aperture greater than 1.38 (Table 1). The condition is expressed in the equation listed above and following:
where NA is the numerical aperture of the objective lens, n is the refractive index, and q is the one-half the objective angular aperture.
TIR Angles and Maximum Angles for Objective Lenses
|
|||||||||||||||||
Table 1
The excitation light must pass through the portion of the lens' numerical aperture cone that is greater than 1.38. Using a high-performance plan apochromatic 1.4 numerical aperture objective, a very small fraction of the lens numerical aperture (1.4 – 1.38 = 0.02) can be utilized. While that is possible, it is very challenging to align the illuminating laser. Even with perfect alignment, increasing the light intensity is difficult because of the miniscule numerical aperture margin available. It is obvious that a lens like the APO 100x/1.65 has a much greater numerical aperture latitude (1.65 – 1.38 = 0.27), making it easier to introduce the excitation light through the objective lens exit pupil (Figure 4 and Table 1).
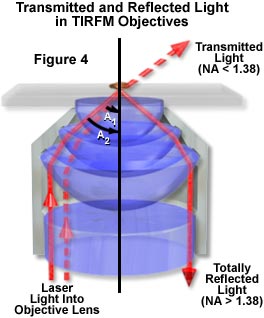
In order to maintain the numerical aperture and achieve the image quality the objective is capable of, a special immersion medium of high refractive index (immersion liquid [Cargille Laboratories, Cedar Grove, NJ]: n(d) = 1.78) must be used. (This is volatile and should be handled using Good Laboratory Industrial Practice).
Also, the cover glass must have a high refractive index (n(d) = 1.788; available from Olympus America Inc.), however its very low volume of manufacture makes it comparatively costly.
Additional Applications
The lens has benefits for other applications as well. Good near-infrared transmission makes it useful for laser trapping (transmission at 1064 nanometers equals 60 percent). A specialized prism is available for super-resolution DIC and capitalizes on the objective lens numerical aperture.
Fluorescence microscopy gains a more than 30 percent intensity advantage over the 100x/1.4 numerical aperture objective. The techniques of TIRFM, DIC, and standard fluorescence microscopy can also be combined.
Configuration
The authors' setup for TIRFM (details given in Tables 2 and 3) uses the IX70 inverted microscope (Olympus America Inc.) equipped to include the right-side camera port and IX laser port. The laser is located to the left of the microscope. Through the 180-millimeter laser port tube lens, a laser is projected into the exit pupil of the illuminating/imaging lens (see Figure 5).
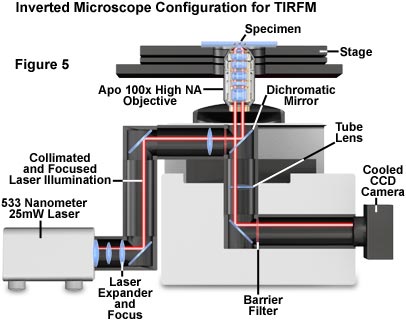
As a light source, a low-noise 20-milliwatt solid-state laser, emitting at 532 nanometers (Crysta Laser, Reno, Nevada), was used. Argon, helium-neon, and krypton lasers can also be used, either coupled directly or via optical fiber.
The limited image intensity achieved with TIRFM suggests the use of a cooled monochrome camera such as the OLY-110, OLYMPIX 2000, or MagnaFire (Olympus America Inc.).
Sample Setup for IX70 TIRFM
(All products from Olympus America Inc.)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2
Recommended Third-Party Hardware/Materials
|
|||||||||||||||||
Table 3
Imaging experiments
On a cover glass, a specimen consisting of 0.2-µm fluorescent beads and tetra methyl rhodamine isothiocyanate (TRITC) stained cheek cells were prepared, and the fluorescence image and the TIRFM image were compared (see Figures 6 and 7).
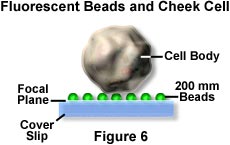
In fluorescence mode, having focused on the beads, the bead fluorescence is very difficult to distinguish because of the obscuring background fluorescence from the cheek cells. In TIRFM imaging mode, the image contrast is dramatically high, and beads can be observed easily.

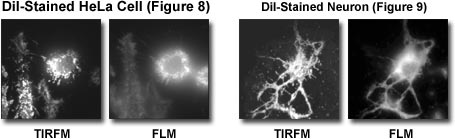
Only the near field (a few hundred nanometers below the specimen side coverslip surface) is excited. The cheek cells are out of reach of the evanescent wave and do not contribute to the fluorescence image. In other experiments, DiI-stained Hela cells and neurons were observed, and fluorescence and TIRFM were compared. In each case, TIRFM permitted very high contrast observation of the cell surface (Figures 8 and 9).
Conclusions
The APO 100x/1.65 is a very high resolution objective that is offered at a time when interest in TIRFM is increasing. The high numerical aperture of the objective, convenient IX70 configuration, and relative economy of solid-state lasers bring TIRFM into the mainstream of microscopy imaging techniques. Application of the objective in conjunction with TIRFM offers new and exciting investigative opportunities to the research community.
References
Tokunaga M., Yanagida T., Single molecule imaging of fluorophores and enzymatic reactions achieved by objective-type total internal reflection fluorescence microscopy, Biochemical and Biophysical Research Communications: 235, 47–53 (1997).
Axelrod D., Total internal reflection fluorescence at biological surfaces, in Noninvasive techniques in cell biology, Wiley-Liss, New York, New York (1990).
Steyer J. and Almers, W., Transport, docking and exocytosis of single secretory granules in live chromaffin cells, Nature: 388, 474 (1997).
Haugland R., (ed) Handbook of Fluorescent Probes and Research Chemicals, 6th ed., Molecular Probes Inc., Eugene, Oregon (1997).
Contributing Authors
Y. Kawano and R. G. Enders - Olympus America, Inc., Two Corporate Center Drive, Melville, New York, 11747.
BACK TO TIR FLUORESCENCE MICROSCOPY
