Fluorescence Digital Image Gallery
Rat Jejunum Myenteric Plexus Enteroglial Cells (EGC/PK060399egfr)
|
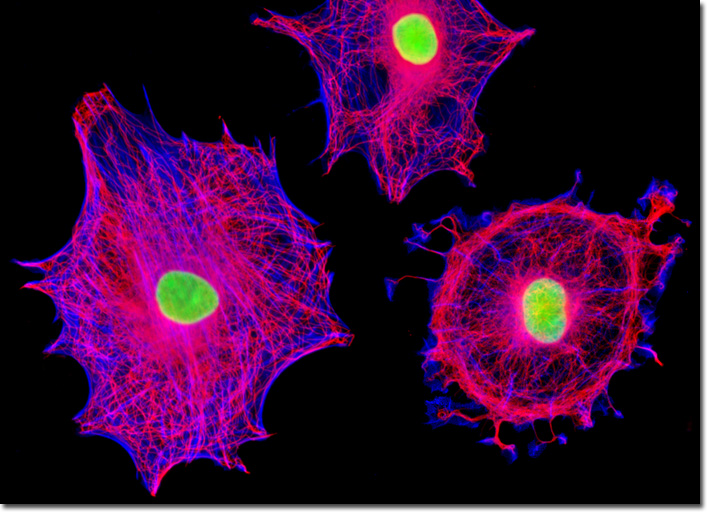
Tubulin is a protein that comprises the basic structural subunits of microtubules, the cytoskeletal filaments associated with the positioning of membrane-bound organelles and intracellular transport. Each tubulin subunit is, in fact, a heterodimer composed of alpha-tubulin and beta-tubulin components linked to one another by noncovalent bonds. The two globular proteins, which only occur in this network, each exhibit a binding site for a single guanosine triphosphate (GTP) molecule. The GTP molecule bound to the alpha-tubulin monomer may be regarded as a fundamental component of the tubulin heterodimer complex because it is physically unable to be exchanged or hydrolyzed, instead remaining constantly confined at the dimer interface. The GTP nucleotide in the beta-tubulin monomer, however, is exchangeable and may be hydrolyzed to produce guanosine diphosphate (GDP), an event that significantly impacts the dynamics of the microtubular network. The rat enteroglial cell culture presented above was immunofluorescently labeled with mouse anti-alpha-tubulin primary antibodies followed by goat anti-mouse secondary antibodies conjugated to Alexa Fluor 568. In addition, the specimen was stained with Alexa Fluor 350 conjugated to phalloidin and SYTOX Green, targeting the filamentous actin network and nuclei, respectively. Images were recorded in grayscale with a QImaging Retiga Fast-EXi camera system coupled to an Olympus BX-51 microscope equipped with bandpass emission fluorescence filter optical blocks provided by Omega Optical. During the processing stage, individual image channels were pseudocolored with RGB values corresponding to each of the fluorophore emission spectral profiles. |
© 1995-2025 by Michael W. Davidson and The Florida State University. All Rights Reserved. No images, graphics, software, scripts, or applets may be reproduced or used in any manner without permission from the copyright holders. Use of this website means you agree to all of the Legal Terms and Conditions set forth by the owners.
This website is maintained by our
|