|
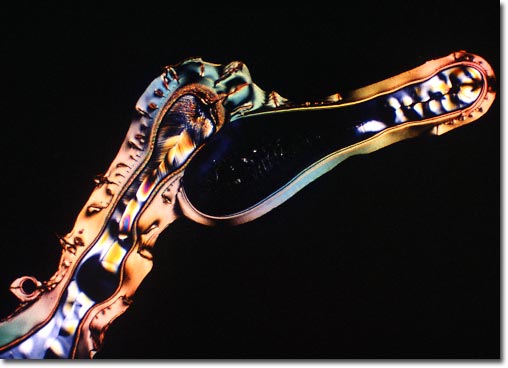
Ascorbic acid is a fragile molecule that is easily destroyed in neutral or alkaline solutions, at elevated temperatures, and upon exposure to oxygen. The vitamin C structure is freely soluble in water, less so in alcohol, and insoluble in certain chemicals such as chloroform, ether, and benzene. In its natural state, ascorbic acid appears in the form of a white to yellowish crystal or powder. The chemical name ascorbic acid refers to L-ascorbic acid, the levorotatory isomer, and has been widely synthesized as a supplement or food additive. Fruits and vegetables are rich natural sources, however the vitamin C content can be rapidly depleted as food becomes stale or when the labile biochemical leaches into water during cooking. The photomicrograph presented above was made with vitamin C recrystallized from the melt and has been titled: Vitamin C Horse.
|